Background

An orange can be used to power a digital clock. An example of galvanic cell!
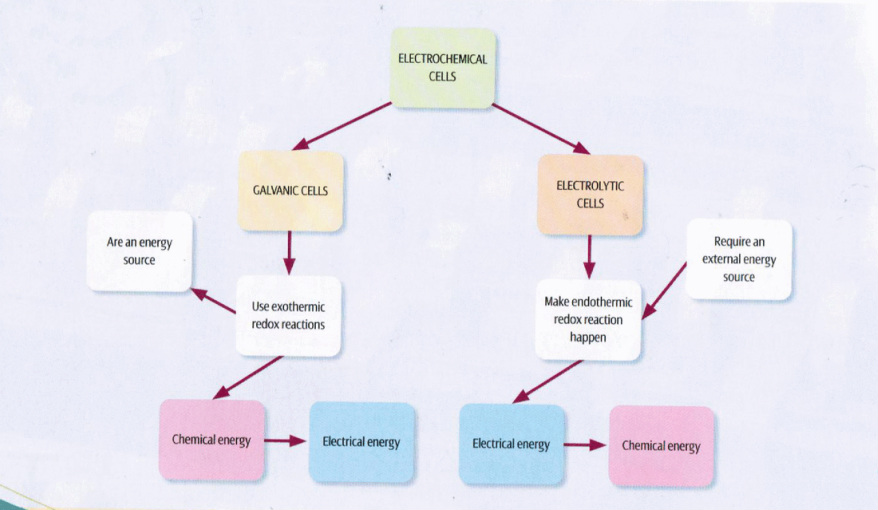
Electrochemistry is the study of chemical reactions which take place in a solution either at the interface of a conductor or within an ionic solution which is called the electrolyte.
Over the next few pages we would be considering two types of cells; the galvanic cell and the electrolytic cell. Within each cell there will be detailed information and description of a general and specific cell associated with the respective cells.
The purpose of these cells is to be able to identify the essential concepts of galvanic and electrolytic cells and how they are used in today's society.
Note: Both of these cells are considered as electrochemical cells.
The specific galvanic cell that will be undertaken is lead acid battery.
The specific electrolytic cell that will be undertaken is Aluminium production.
Over the next few pages we would be considering two types of cells; the galvanic cell and the electrolytic cell. Within each cell there will be detailed information and description of a general and specific cell associated with the respective cells.
The purpose of these cells is to be able to identify the essential concepts of galvanic and electrolytic cells and how they are used in today's society.
Note: Both of these cells are considered as electrochemical cells.
The specific galvanic cell that will be undertaken is lead acid battery.
The specific electrolytic cell that will be undertaken is Aluminium production.