General Review
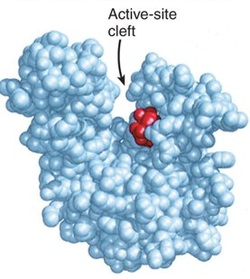
A typical enzyme: red is the active site
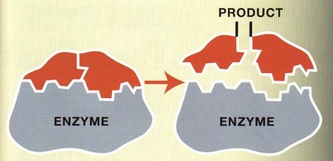
Enzymes are proteins which act as biological catalyst or in simple words, its a molecules which increases reaction rates within the body or elsewhere. Enzymes are highly specific, one enzyme will catalyze and act on one specific type of substance. The human body is a complex 'factory' one which has to be highly regulated to function properly. Enzymes are responsible in carrying out the regulation of the body. They are involved in every reaction of life since they help them to occur faster. Enzymes also control the manufacture of complex substances such as skin and blood, as well as the breaking down of chemicals to provide energy. In another word enzymes make life possible.
Structure
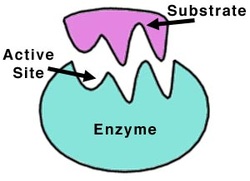
A simple representation of an enzyme
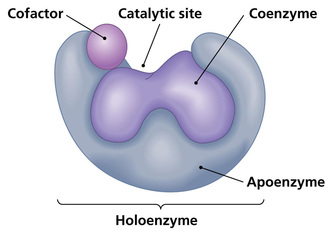
Just like all proteins, enzymes follow the same rules like they are long, linear chains of amino acids that go through all the different kinds of coiling and folding to produce a 3D structure. Every amino acid sequence is unique which in turn gives that enzymes a specific structure. Enzymes in general are globular proteins and they themsevles are much larger than the substrates that they act on. Substrates are substances which the enzymes acts on, the enzyme can break it down or bring two substrates together to form a larger molecule. The binding of the substrate happens in the most important location of the enzyme, its the active site of the enzyme. The active site of an enzyme is found in a cleft or pocket that is lined by amino acid residues ( otherwise known as nucleotides in ribozymes, a RNA enzyme which enables it to catalyze a reaction), these recognizes the substrate and commences the chemical reaction afterwards. A series of bonds and interactions such as hydrogen bonds, hydrophobic interactions, temporary covalent interactions (van der Waals forces) or a combination of all of these eventually form the enzymes-substrate complex. Within the enzymes there may be cofactors, these are non-protein chemical compounds which can be considered as 'helper molecules' which aid the enzyme in biological transformations. Cofactors can either be organic or inorganic with the less loosely bound cofactor associated with the enzyme is called a coenzyme. If an inactive enzyme is without a cofactor, its called an apoenzyme conversely an enzyme with a cofactor is called a holoenzyme.
Function
Enzymes have specific 3D structure
The overall function of an enzyme is to speed a reaction or catalyze it really quickly. Consider the enzyme urease which is found in airborne bacteria, they catalyze urea, one urease enzyme can catalyze 30,000 urea molecules per second! Now without the urease, the urea to decompose by itself would take about 3 million years! Hence this is why we need enzymes to do something really fast. As well as this, enzymes are highly specific meaning that they will catalyze only those which can fit inside its active site. An enzymes works by lowering the activation energy which is needed to kick start a reaction. Enzymes are really sensitive molecules with narrow range of conditions for functioning. There are several factors which can affect the function of an enzyme, these are temperature, pH, enzyme concentration and substrate concentration.
Temperature
At low temperatures the activity of the enzyme is low but as the temperature increases the activity or rate of reaction also increases. This is so because at higher temperatures the molecules absorb more energy and hence collide more with the enzyme so consequently more reaction occurs. If the temperature exceeds the limit that its high enough to damage the enzyme's structure, the function of the enzyme ceases and the enzyme is said to be denatured (a permanent change in its structure).
pH
Extremes in acidity (pH) will also cause the enzyme's structure to denature. The active site has an electric charge to it, if the environment becomes too acidic or basic the active site's charge may change. Since the charge changes, a lot of the interaction and bonds hang in the balance. As each types of bonds became more or less attracted or repelled because of the changing charges, the structure eventually changes. Not to mention that they 3D structure of a protein is due to all the different types of interactions and bonds.
Enzyme concentration
What this really means that if there an increasing concentration of enzymes with ample substrates and cofactors, the rate of reaction will increase in a linear straight line.
Substrate concentration
This means that if there an increasing concentration of substrates but a fixed amount of enzyme with ample cofactors, the rate of reaction will increase but continue at a constant rate of reaction. The graph of this type of factor influencing the enzyme activity is an inverse parabolic shape but continuing in a horizontal straight line from the turning point. This is because the enzymes would be filled or saturated with substrate at a point and the rate of reaction would be constant from that point onwards (see illustrations below)
Temperature
At low temperatures the activity of the enzyme is low but as the temperature increases the activity or rate of reaction also increases. This is so because at higher temperatures the molecules absorb more energy and hence collide more with the enzyme so consequently more reaction occurs. If the temperature exceeds the limit that its high enough to damage the enzyme's structure, the function of the enzyme ceases and the enzyme is said to be denatured (a permanent change in its structure).
pH
Extremes in acidity (pH) will also cause the enzyme's structure to denature. The active site has an electric charge to it, if the environment becomes too acidic or basic the active site's charge may change. Since the charge changes, a lot of the interaction and bonds hang in the balance. As each types of bonds became more or less attracted or repelled because of the changing charges, the structure eventually changes. Not to mention that they 3D structure of a protein is due to all the different types of interactions and bonds.
Enzyme concentration
What this really means that if there an increasing concentration of enzymes with ample substrates and cofactors, the rate of reaction will increase in a linear straight line.
Substrate concentration
This means that if there an increasing concentration of substrates but a fixed amount of enzyme with ample cofactors, the rate of reaction will increase but continue at a constant rate of reaction. The graph of this type of factor influencing the enzyme activity is an inverse parabolic shape but continuing in a horizontal straight line from the turning point. This is because the enzymes would be filled or saturated with substrate at a point and the rate of reaction would be constant from that point onwards (see illustrations below)
Types of models
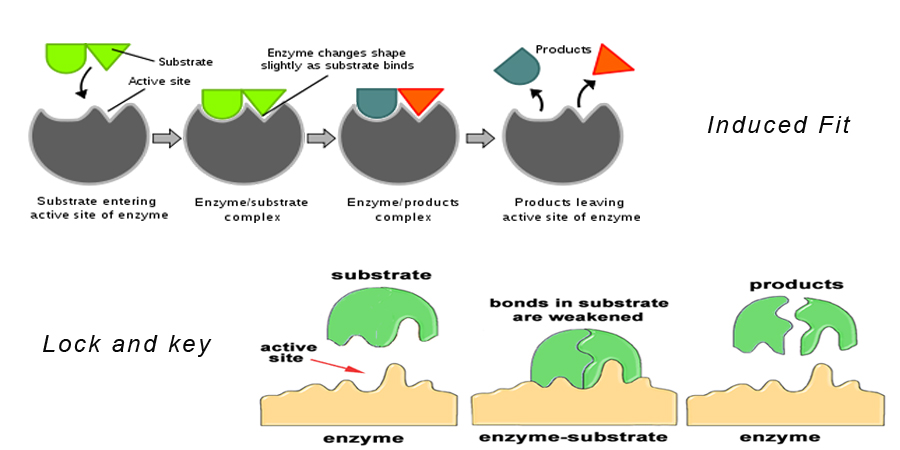
Lock and Key
This earlier model describes substrates being drawn into clefts where the shapes are complementary meaning its like a jigsaw puzzle, the substrate will fit exactly into the active site of the enzyme.
Induced fit model
An enzyme fits to its substrate somewhat like a lock and key. The shape of the enzyme changes when the substrate fits into the cleft. The reactants become bound to enzymes by weak chemical bonds. This binding can waken bonds within the reactants themselves, allowing the reaction to proceed more readily.
This earlier model describes substrates being drawn into clefts where the shapes are complementary meaning its like a jigsaw puzzle, the substrate will fit exactly into the active site of the enzyme.
Induced fit model
An enzyme fits to its substrate somewhat like a lock and key. The shape of the enzyme changes when the substrate fits into the cleft. The reactants become bound to enzymes by weak chemical bonds. This binding can waken bonds within the reactants themselves, allowing the reaction to proceed more readily.
Denaturation of Enzymes
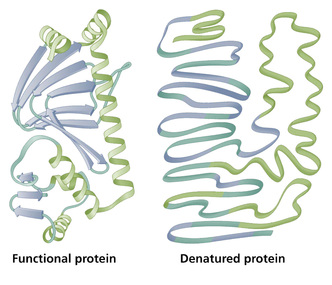
Denaturation is a permanent change in the enzyme's structure, its an irreversible reaction. It is a result of increased temperature or a change in pH or the addition of various chemicals. Once a protein has been denatured, the unfolded chains tend to form randomly looped structure which come into close contact. The chains become entangled and bond with each other, often by disulfide bridges, so large clumps of protein molecules are formed. This is called coagulation. Examples of denaturation is a cooked egg.