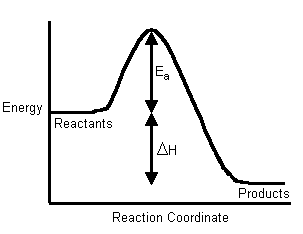
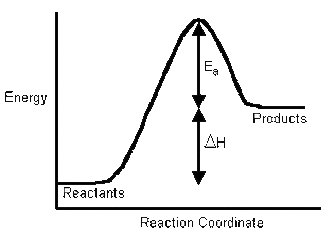
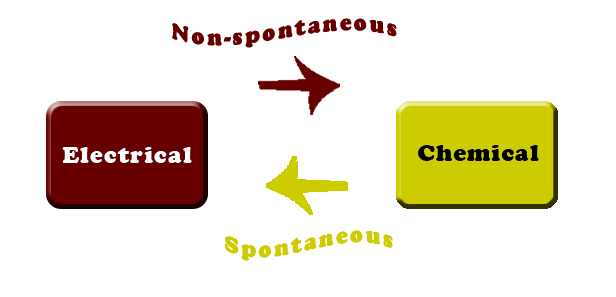
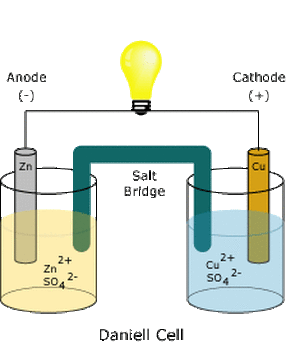
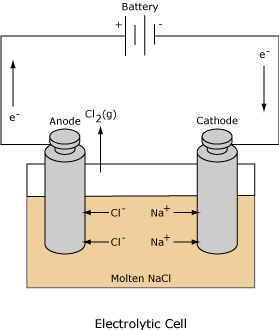
Galvanic and Electrolytic cell
These cells have a lot in common but there are also significant differences as well. The similarities include:
Similarities
Galvanic and electrolytic cell are similar in the sense that they are both:
- Electrochemical cells since energy is being converted to electrical energy or vice versa.
- Both involve oxidation and reduction
- The electrochemical series can be used to predict the reactions in both
- and both has a specific anode and cathode.
Differences
|
|
Bibliography
Books
Gordon J, 2007, Nelson Chemistry, 2nd edition, Nelson school, South Melbourne, Victoria
Commons C, Jarrett S, McKenzie C, Moseley W, Porter M & Williamson M, 1991, Chemistry two, 3rd edition, Harcourt Education, Port Melbourne, Victoria
Hogendoorn B, Commons C, Jarret S, McKenzie C, Moseley W, Porter M & Williamson M, 2007, Chemistry 2, 4th edition, Pearson Education Australia, Port Melbourne, Victoria.
O'Shea Pat, Lisachem 12 Chemistry, Learning Materials, Hampton East, Victoria.
Online sources
A Pearson Company, 2004, Prentice-Hall Inc, viewed on 19 September 2012, http://wps.prenhall.com/wps/media/objects/602/616516/Chapter_18.html
From Wikipedia the free encyclopedia, 2012, viewed on 19 September 2012, http://en.wikipedia.org/wiki/Galvanic_cell
From Wikipedia the free encyclopedia, 2012, viewed on 19 September 2012, http://en.wikipedia.org/wiki/Electrolytic_cell
Pictures
Figure 1, 3, 5 & 10
Nelson Chemistry, Nelson school,
Figure 2 and 3
General Chemistry: Principles, Patterns, and Applications, v. 1.0, http://catalog.flatworldknowledge.com/bookhub/4309?e=averill_1.0-ch19_s02
Figure 6
Flash Physics animation, http://www.wainet.ne.jp/~yuasa/EngF2.htm
Figure 7 and 8
Lead acid battery, http://en.wikipedia.org
Figure 11
MANUFACTURING CHLORINE USING A DIAPHRAGM AND A MEMBRANE CELL, http://www.weebly.com/weebly/main.php
Figure 12
Membrane cell, http://www.eurochlor.org/the-chlorine-universe/how-is-chlorine-produced/the-membrane-cell-process.aspx
Gordon J, 2007, Nelson Chemistry, 2nd edition, Nelson school, South Melbourne, Victoria
Commons C, Jarrett S, McKenzie C, Moseley W, Porter M & Williamson M, 1991, Chemistry two, 3rd edition, Harcourt Education, Port Melbourne, Victoria
Hogendoorn B, Commons C, Jarret S, McKenzie C, Moseley W, Porter M & Williamson M, 2007, Chemistry 2, 4th edition, Pearson Education Australia, Port Melbourne, Victoria.
O'Shea Pat, Lisachem 12 Chemistry, Learning Materials, Hampton East, Victoria.
Online sources
A Pearson Company, 2004, Prentice-Hall Inc, viewed on 19 September 2012, http://wps.prenhall.com/wps/media/objects/602/616516/Chapter_18.html
From Wikipedia the free encyclopedia, 2012, viewed on 19 September 2012, http://en.wikipedia.org/wiki/Galvanic_cell
From Wikipedia the free encyclopedia, 2012, viewed on 19 September 2012, http://en.wikipedia.org/wiki/Electrolytic_cell
Pictures
Figure 1, 3, 5 & 10
Nelson Chemistry, Nelson school,
Figure 2 and 3
General Chemistry: Principles, Patterns, and Applications, v. 1.0, http://catalog.flatworldknowledge.com/bookhub/4309?e=averill_1.0-ch19_s02
Figure 6
Flash Physics animation, http://www.wainet.ne.jp/~yuasa/EngF2.htm
Figure 7 and 8
Lead acid battery, http://en.wikipedia.org
Figure 11
MANUFACTURING CHLORINE USING A DIAPHRAGM AND A MEMBRANE CELL, http://www.weebly.com/weebly/main.php
Figure 12
Membrane cell, http://www.eurochlor.org/the-chlorine-universe/how-is-chlorine-produced/the-membrane-cell-process.aspx