General Review
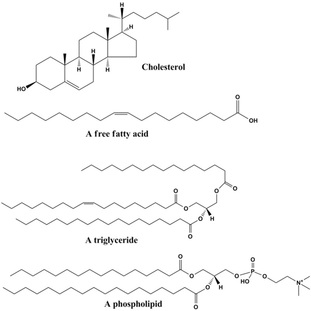
Showing some forms of lipids
Let's be conscious of the fact that lipids are not fats only! But actually fats are a sub groups of lipids. Lipids constitute a very vast range of naturally occurring molecules, these include waxes, fats, sterols (also known as steroid alcohol, a sub group of steroids, these occur naturally in plants, animals and fungi, with the best known being cholesterol) , fat soluble vitamins (such as vitamins A, D, E and K), phospolipids, triglycerides, diglycerides and monoglycerides. To some extents humans and other mammals can synthesis lipids and break them down, there are however some which cannot be synthesised in the body and therefore must be obtained from our diet. All lipids are non-polar and hydrophobic molecules. The main purpose of lipids in biology is to provide energy storage, as structural components of cell membranes and as improtant signaling molecules. Lipids are broken down into further 8 sections and these are fatty acids, glycerolipids (glycerol), glycerophospholipids (a fancy name for phospholipids), sterol lipids, prenol lipids, polyketides, sphingolipids and saccharolipids. However I would just focus on the first 4 lipids as these are the most common and are taught in high schools and maybe first year university.
Sources of fat

Sources of fats
Even though we make most of our fats internally, fats make up a lot of our daily life diets. Examples are like butter, whole milk, ice cream, cream, cheese and fatty meats. Fats are sometimes found in some vegetable oils like coconut, palm, and palm kernel oils. We tend to enjoy fatty foods because they remain in our stomach for longer e.g they get oxidised or burnt less quickly than glucose even though fats stores about twice as much energy as carbohydrates. Fats provide about 9 kilocalories per gram and carbohydrates provide about 4 kilocalories per gram. Since fats remain for longer in our stomach it gives us the sensation of being full or satisfied.
Fats and oils
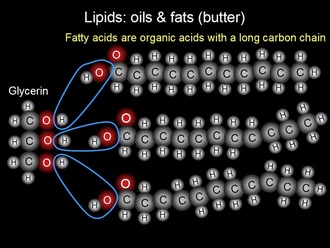
Fats and oils are compromised of a single molecule of glycerol and three molecules of fatty acid. Fatty acids contain a carboxy functional group (COOH, is an acid hence their name.) The real purpose of fatty acids are that when they are metabolized or burnt, they yield large quantities of ATP. Within our body, different type of cells prefer either glucose of fatty acids. One in particular is the skeletal muscles and the heart which prefers fatty acids over glucose whereas the brain relies upon glucose of ketone bodies (other monosaccharides.) The body sometimes cannot synthesis fatty acids therefore these are called essential fatty acids which must be obtained from our diets. Examples of this essential fatty acids are linoleic acid (LA) and alpha-linolenic (ALA) which are widely spread out in vegetable oil. The glycerol molecule contains a hydroxy (OH) functional group for this reason they are said to be an alcohol group. Fats and oils are made from three molecules of fatty acid and one molecule of glycerol which under goes condensation reaction to from a triglyceride or tristearin which is a fat molecule and three water molecules. The reaction between these two molecules is called esterification because the reaction forms an ester functional group (-COO-). Triglycerides are large non-polar molecules and hence insoluble in water.
What separates fats from oils is their properties which is that fats are solids and oils are liquids at room temperatures. Otherwise they are basically the same thing.
What separates fats from oils is their properties which is that fats are solids and oils are liquids at room temperatures. Otherwise they are basically the same thing.
Types of fats and oils
There are a wide range of fats and oils, each one with its distinctive properties.
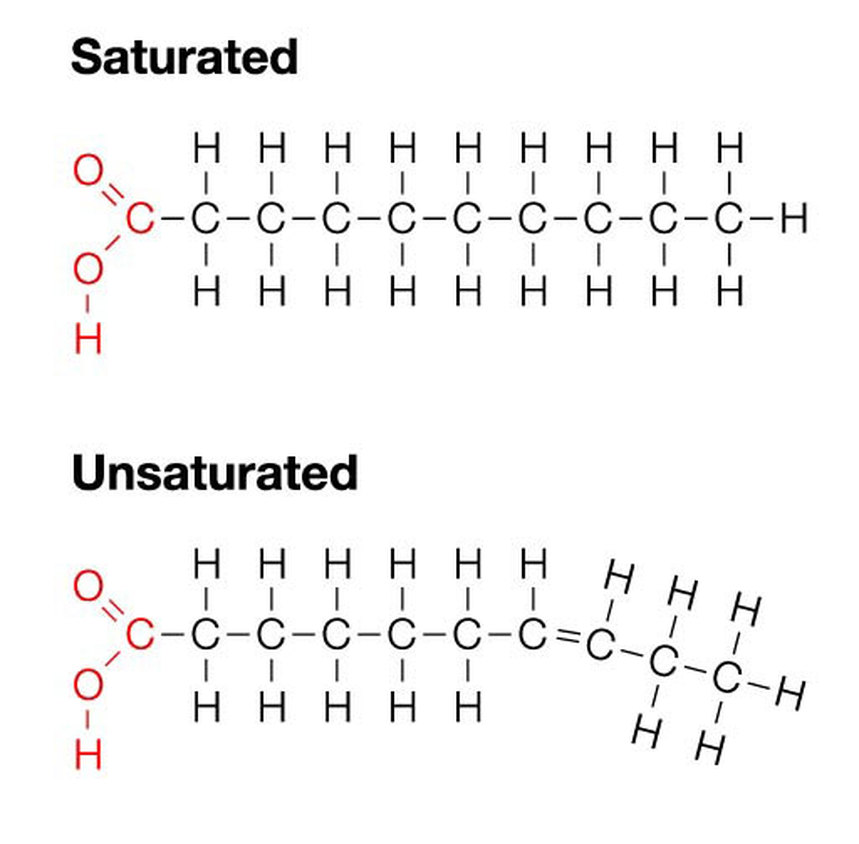
SaturatedThese types of fats contain only single carbon-carbon bonds. They have the maximum number of hydrogen atoms and does not contain any kinks. These are normally fats and hence solids at room temperatures. Saturated fats pack more closely and hence resulting in a stronger dispersion forces. Therefore they have higher melting points and are solids.
|
Mono-unsaturatedThese ones contain at least one carbon-carbon double bond and therefore has a kink within their structure. Mainly these types of lipids are oils and therefore liquids at room temperatures.
|
PolyunsaturatedThese are all oils since they do not pack closely and has more than one double carbon-carbon bond resulting in more than one kinks and as a result, they have lower melting points and are liquids at room temperatures.
|
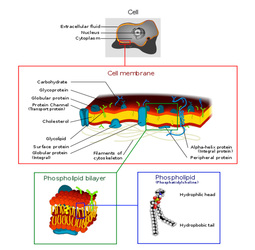
Phospholipids
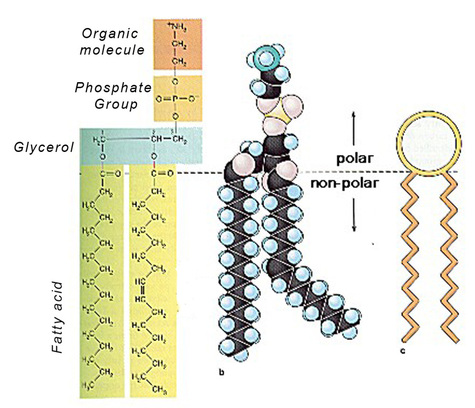
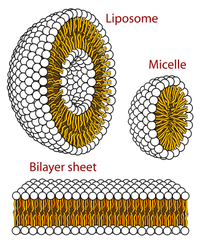
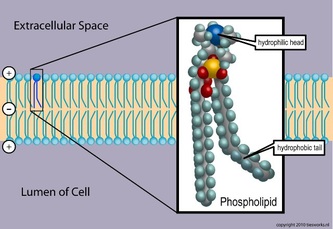
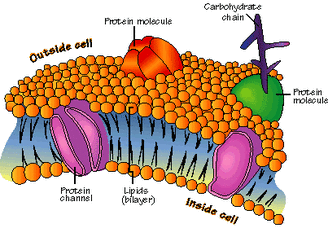
Phospholipids are also a class of lipids. Its composed of a diglyceride, a phosphate group and a simple organic molecule. The diglyceride is one glycerol molecule attached to two fatty acid molecules. Phospholpids are of large importance to us as they make up a major component of living organism's cell membranes because they have the potential to form lipid bilayers. The structure of a phospholipid molecule is a head being hydrophilic (water loving, polar group) and a tail being hydrophobic (water hating, non-polar group). The head of the phospholipid contains the negatively charged phosphate group and the tail is composed of the long fatty acid molecule. A unique property of phospholipid is when they are placed in the water, they form different structure depending on the specific properties of the phospholipid itself. For example they may form a liposome or a micelle or a bilayer. (see illustrations below.)The lipid bilayer is very unique in that it stops ions, proteins and other molecule from random diffusion. It is impermeable to water soluble substances and to charged ions like salt. This is really important in maintaining a stable body. The phospholipid bilayer is compromised of of two thin sheets of phospholipid and they arrange themselves by their two tails touching each other. Having a structure like this only allows for lipid soluble molecules to readily dissolve. There are also other molecules and substances that are associated with phospholipid bilayer, these are protein channels, glycolipids, cholesterol and a other molecules. Each one has a specific function that helps the bilayer work properly.