General Review
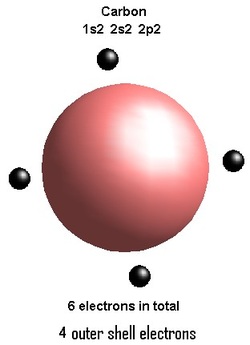
Outer shell of a Carbon atom
The empirical formula of a carbohydrate molecule is Cn(H2O)n, composed of Carbon, Hydrogen and Oxygen in the ratio of 1:2:1. There is however an exception to this rule as is with every rule and that is that the formula of deoxyribose sugar, which is a component of DNA, the stuff that makes you tick; has a formula of C5H10O4. Carbohydrate is a branch of organic compounds. Organic compounds are those that typically contain within their structures carbon atoms. Carbon is the most fundamental element in which it has the capability to form thousands of long chains or polymers. If you look at the structure of a carbon molecule, you will see that it has 4 outer electrons, its electronic shell configuration is 2,4 and it sub-shell configuration is 1s2 2s2 2p2, in each case you will observe that it has the capacity to take four more electrons to form a stable outer shell. Hence a carbon molecule has the potential to form four covalent bonds with other atoms, mainly with those which can potentially form a stable outer shell as well like Hydrogen. This property of carbon is curcial to organic chemistry. Carbohydrates are viewed as hydrates of carbon hence their name carbohydrates. Hydrates simply means that the substance contains water molecules in them. This is critical in building or breaking down carbohydrates otherwise know as condensation and hydrolysis reactions. In Biology the study of organic compounds mainly carbohydrates, fats and proteins are called Biomacromolecules and in Chemistry its called Biochemistry. Carbohydrates are really important in our diet because they provide us with the main energy source which drives our daily lifestyles in the form of glucose which then goes through oxidation process to give us ATP (Adenosine triphosphate) or energy. Let's now look at various aspects of carbohydrates!
Sources of Carbohydrates

Major sources of Carbohydrates
Main sources of carbohydrates include whole grain (wheat, rye, barley, rice, millet, buckwheat and oats) whether they are whole or refined, raw or cooked. Legumes are also a good source of carbohydrates they include peanuts, lentils, beans and peas. Some vegetable that contain mild carbohydrates are carrots, cauliflower, beets, rutabaga and salfiy. Vegetables that contains abundant amounts of carboydrates include pumpkin, caladium root, taro root, cassava root and Jerusalem artichokes. Also not mention fibers such as breads which delivers a lot of carbohydrates and are very good in helping digestion. Another good source of carbohydrates are sweet natural fruits which contains sugars like glucose or fructose such as in honey, mangoes or apricots. Its immensely important to have a very balanced diet which is crucial to the maintenance of a healthy livelihood. These vegetables, fibers and fruits promote a healthy body by delivering essential minerals, vitamins and a host of nutrients, all of which is very useful to the body.
Monosaccharides
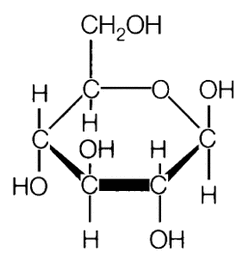
A glucose molecule
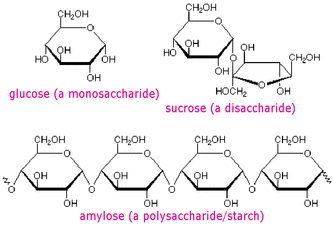
Monosaccharides are referred to as simply sugars, they are monomers to more complex carbohydrates and the simplest of carbohydrates. Originally the word 'saccharide' was derived from the Greek word σάκχαρον (sákkharon), meaning "sugar". A typical monosaccharides contains an aldehyde (R-CH=O, where R represents an alkyl group or a side chain) or ketone ( RC=OR, with R representing an alkyl or group of atoms) and with many hydroxy (OH) functional groups. Both ketone and aldehyde are organic compounds and their functional group is called the carbonyl group (a carbon double bonded to oxygen, see the illustrations below). Monosacchardies include glucose, fructose and galactose, monosaccarides with 3 carbon atoms are called triose, with four carbon atoms are called tetroses, five are called pentoses, six are called hexoses and so on. Any structure with five or more carbon atoms will form a ring like glucose. Each of the monosaccharides all have the same composition e.g their formula is C6H12O6 but they are structurally different. For example glucose is an aldohexose (a six-carbon aldehyde), fructose is a ketohexose (a six-carbon ketone) and galactose is an epimer of glucose meaning that they differ in one of the carbon atom (see illustration bellow). These differences in their structure's are small but are of great importance to living organisms. As you can from the structural formulas of these monosaccharides, they contain hydroxy (OH) polar groups which enables them to form hydrogen bonds with water molecules and dissolve hence thats why they are really soluble in water. Among the monosaccharides, glucose is found to be abundant; its found in all living things. It is also a major component of most of the larger carbohydrates like cellulose and starch. On the other hand fructose and galactose are not so abundant as glucose, in fact galactose in not found by itself in nature at all but found as a component of larger carbohydrates. Fructose is also a component of other larger carbohydrates and is the sweetest of sugars known, being 1.7 times sweeter than table sugar.
Disaccharides

Normal day sugar is sucrose C12 H22 O11
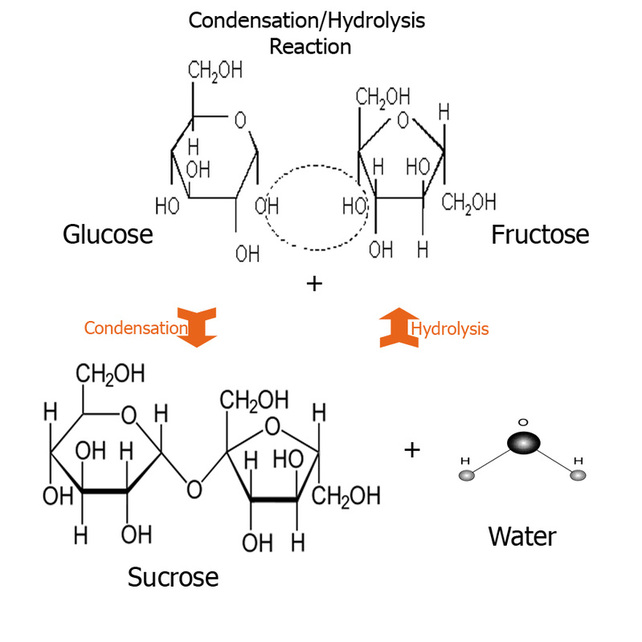
Disaccharides means two sugars, its the next simplest sugar after monosaccharides. Examples of these are maltose formed by the condensation reaction between glucose and glucose molecules. Maltose also serves as compound alongside glucose and other sugar to be fermented and produces ethanol in absence of oxygen (absence of oxygen is known as anaerobic respiration.) Then there is sucrose which is formed between glucose and a fructose molecules and lastly lactose which is found in milk; is formed between glucose and a galactose molecules. In here the monosaccharides can be considered to be building block of the more complex carbohydrates like polysaccharides, remember that disaccharides are still considered to be simple sugars, its only the polysaccharides which are long chains and complex compounds. Condensation reaction is one where the hydroxy group (OH) of one monosaccaride reacts with an OH group of another monosaccharide and thus forms water. The two rings are joined via an oxygen atom. This linkage is called a glycosidic linkage. This is how more complex carbohydrates than disaccharide are formed e.g polysaccharides
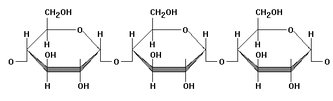
Polysaccharides
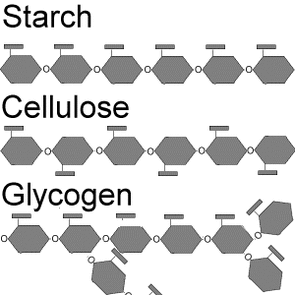
Structural differences of Polysaccharides
Polysaccharides are polymer carbohydrates made by linking monosaccharides into a long chain. These polymers are mainly made up of glucose molecules linked together in different ways by condensation reactions. Polysaccharides are generally insoluble since they are massively long and not mention tasteless. The most three important polysaccharides are:
The roles of these polysaccharides are either storage or structural. For example glycogen acts as a glucose storage molecule in animals. As glucose can quite easily and quickly get oxidised, the body stores reserves of glucose whenever energy is required quickly. Excess of this glucose is polymerised to form glycogen which is stored in the liver and in muscle tissues. When the reserves of glucose are filled up and more glucose is available, its then converted into fat. When energy is required back again, the glycogen is then hydrolysed (opposite to condensation reaction) to yield glucose for cellular respiration.
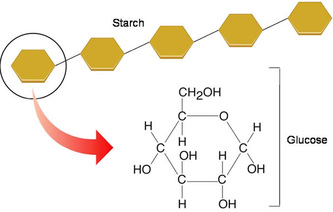
Starch is the equivalent of glycogen but only that this is the storage molecules for plants. The starch is stored and used at night to meet the plant's ongoing energy requirements when glucose production from photosynthesis has ceased.
Cellulose is the main structural material in plants. It is a large linear polymer of glucose. It is slightly different to starch and glycogen because the CH2OH groups on adjacent glucose monomers in starch are on the same side of the polymer chain, while in cellulose they are on alternating sides. In plants individual cellulose molecules are arranged into bundles of parallel chains. These are held together by hydrogen bonding between adjacent chains. Thus these bundles are used to form the cell walls that we know today which encloses the cell and support it.
- Starch
- Cellulose
- glycogen
The roles of these polysaccharides are either storage or structural. For example glycogen acts as a glucose storage molecule in animals. As glucose can quite easily and quickly get oxidised, the body stores reserves of glucose whenever energy is required quickly. Excess of this glucose is polymerised to form glycogen which is stored in the liver and in muscle tissues. When the reserves of glucose are filled up and more glucose is available, its then converted into fat. When energy is required back again, the glycogen is then hydrolysed (opposite to condensation reaction) to yield glucose for cellular respiration.
Starch is the equivalent of glycogen but only that this is the storage molecules for plants. The starch is stored and used at night to meet the plant's ongoing energy requirements when glucose production from photosynthesis has ceased.
Cellulose is the main structural material in plants. It is a large linear polymer of glucose. It is slightly different to starch and glycogen because the CH2OH groups on adjacent glucose monomers in starch are on the same side of the polymer chain, while in cellulose they are on alternating sides. In plants individual cellulose molecules are arranged into bundles of parallel chains. These are held together by hydrogen bonding between adjacent chains. Thus these bundles are used to form the cell walls that we know today which encloses the cell and support it.